EU Reach Compliance
Li Pigments abides to global regulations of permanent makeup and we also abide to EU regulations within the EU.
EU REACH
Li Pigments excels at providing pigments that follow the most current worldwide regulations and guidelines to ensure a high level of safety with minimal health risk for our customers.
Li Pigments abides to global regulations of permanent makeup and we also abide to EU regulations within the EU. With 30+ years of no adverse reactions, our tried & true products are still available. New EU REACH compliant pigments have also been added to our inventory to accommodate our customers and clients within the EU.
The Li Pigments products that cannot be sold within the EU do not contain and will not ever contain any banned raw pigment powders or banned chemicals/carriers.
Any Li Pigments products that are not EU Compliant previously passed the EU-Resolution ResAP(2008)1 with certificate validation.
QUALITY CONTROL
Exact Shades Every Time…
Our modern processing equipment and strict production controls allow us to recreate exact shades every time. The quality control department controls all Standard Operating Procedure (S.O.P.), the release of all materials, finished goods, and the distribution of documents for specifications, test methods, and release criteria.OUR FACILITY
Li Pigments is a Certified Cosmetic Manufacturer headquartered in New Jersey. The facility and warehouse encompasses the administration, production lab, stability quality control, and packaging departments. We have a state-of-the-art production lab equipped to handle formulations and pigment production with the most advanced, modern technologies.Testing & Analysis
Li Pigments adheres to stringent high-quality control standards that follow all cosmetic manufacturing protocols. We’re a state-licensed certified cosmetic establishment certified for micro-pigmentation, permanent cosmetics, and tattooing, as well as traditional cosmetics. All pigments adhere to the highest standards for purity, tested to comply with their respective specifications for use in micro-pigmentation procedures.- Bioburden Batch Testing results show the microbial and toxicity levels of the product before sterilization.
- Total Microbial Count (before sterilization): Less than 20 organisms/gm
- Total Microbial Count (after sterilization): 0 organisms/gm
- Sanitation & Sterilization: Where applicable, various products we manufacture are gamma-sterilized to prevent infection & contamination.
CERTIFICATIONS
- Investigation of Aromatic Amines with Carcinogenic, Mutagenic, Reprotoxic and sensitizing properties according to EU Resolution ResAP (2008)1
- Investigation of Carcinogens classified in Categories 1, 2 and 3 by the European Commission and mentioned in the Council Directive 1967/548/EEC of 27 June 1967 According to EU Resolution ResAP (2008)1 aromatic Amines with Carcinogenic, Mutagenic, Reprotoxic and sensitizing properties according to EU Resolution ResAP (2008)1
- GC/MS-Analysis
- Dyes Causing Allergies
- Mutagenic and Carcinogenic Dyes
- HPLC-Analysis
- Heavy Metals
- PAH’s. Polyaromatic Hydrocarbons and Benzene-a-pyrene (BaP)
- Germ Test, Total Germ Count
- Microbiological Test of Sterility
MANUFACTURING CONTROL GUIDELINES
EU REACH

SUPPLYING QUALITY PERMANENT COSMETICS SINCE 1992
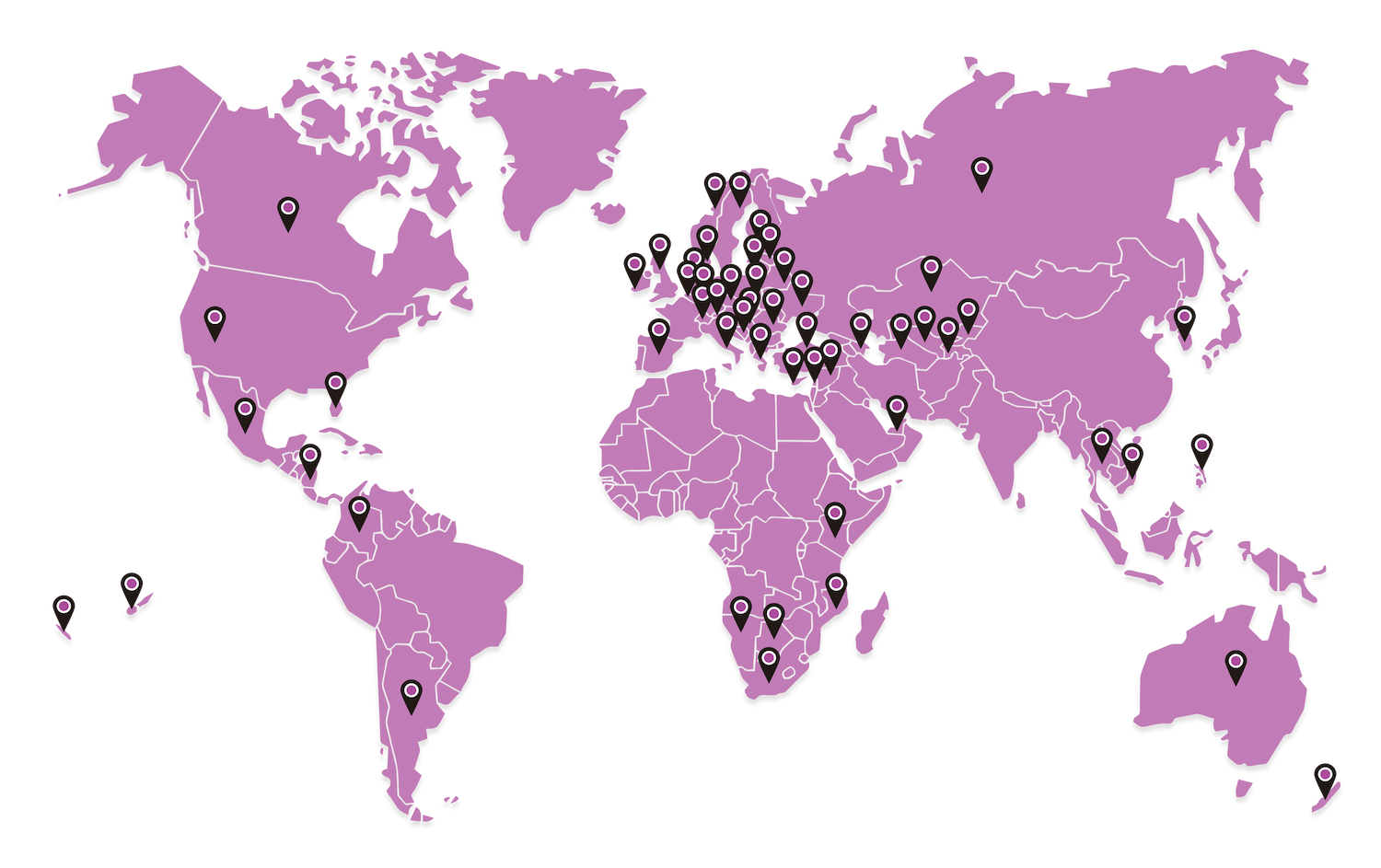
FIND A DISTRIBUTOR